
The history of energy is the story of how a broad philosophical idea gradually became one of the most fundamental concepts in physics. The word “energy” ultimately traces back to the Greek term energeia, used by Aristotle to mean something close to actuality, activity, or being-at-work. In Aristotle’s philosophy, it was not yet the modern measurable physical quantity used in science today.
Big idea: “Energy” evolved from a philosophical description into a precise, conserved numerical quantity that ties together mechanics, thermodynamics, relativity, and quantum theory.
From vis viva to early mechanics
The modern scientific path begins in the late 17th century with Gottfried Wilhelm Leibniz. He introduced the idea of vis viva (“living force”), proportional to a body’s mass times the square of its velocity. It was not identical to the modern formula for kinetic energy, but it was a major step toward it.
In the 18th century, Daniel Bernoulli extended related reasoning to fluids in Hydrodynamica (1738), and Émilie du Châtelet helped clarify that the effect of motion scales with velocity squared rather than velocity alone.

The word “energy” enters physics
The word energy entered physics in its modern mechanical sense with Thomas Young, usually credited from his 1802 lectures and their 1807 publication. Young used “energy” in place of vis viva.
A little later, Gaspard-Gustave de Coriolis introduced the terms work and kinetic energy in their modern meanings in his 1829 work on machines. In 1853, William J. M. Rankine introduced the term potential energy, helping establish the familiar pair of kinetic and potential energy.
Work as the bridge: force through distance
The idea of energy became clearer through the concept of work. In physics, work means exerting a force on a body and making it move through some distance. To lift a weight against gravity is work; to drive a nail into wood against friction is also work.
Anything capable of performing work is said to possess energy. A moving object can do work because of its motion, so it possesses kinetic energy. Heat can do work by expanding steam; electricity can do work in circuits and motors; and light, sound, magnetism, and chemical processes can all be involved in work and energy transfer.
Energy in practice: common forms
- Kinetic: motion
- Potential: position/configuration
- Thermal: heat and temperature
- Electrical & magnetic: fields and circuits
- Chemical: bonds and reactions
- Radiant: light
Conservation thinking: from mass to energy
A useful analogy came from chemistry. In the 18th century, Antoine Lavoisier played a central role in establishing the principle now known as conservation of mass in chemical reactions: matter can change form, but the total mass of reactants and products remains the same.
This encouraged scientists to wonder whether energy might also obey a conservation law. Establishing that law was experimentally difficult because energy was harder to measure and isolate than mass.
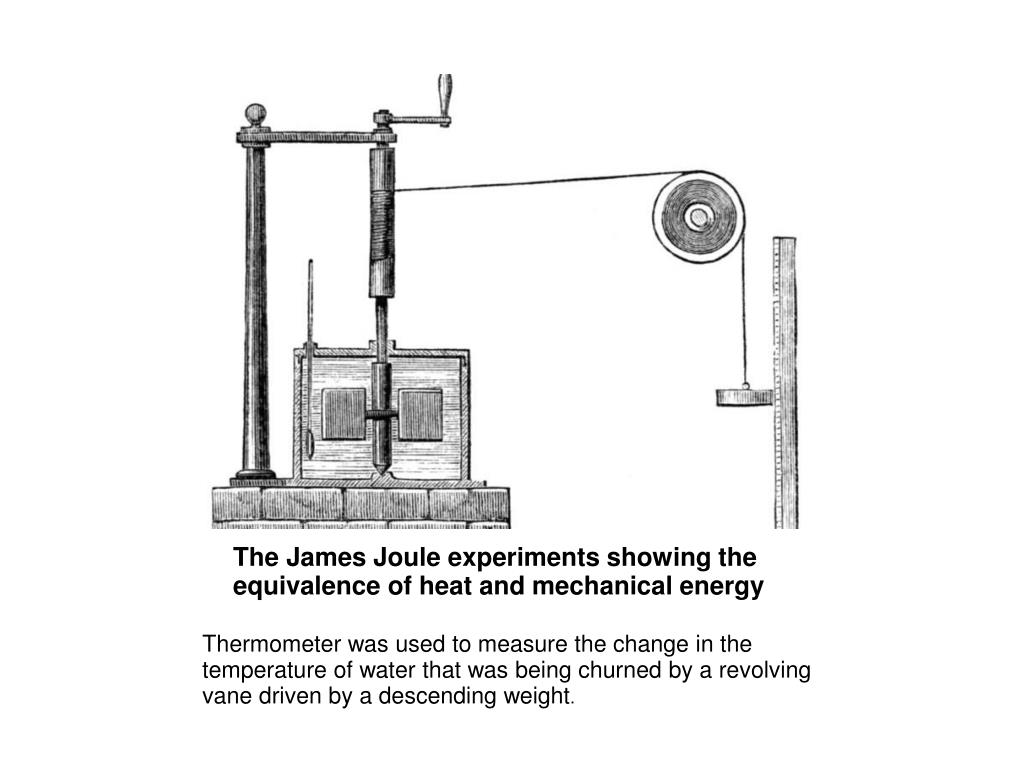
Joule and the mechanical equivalent of heat
Beginning in the 1840s, James Prescott Joule carried out experiments showing that different forms of work could be converted into definite, measurable amounts of heat. This established the mechanical equivalent of heat and helped demonstrate that heat is a form of energy rather than a separate substance.
Around the same period, Julius Robert von Mayer and Hermann von Helmholtz helped formulate the more general principle that energy is conserved. Energy can change form, but not total amount: energy cannot be created or destroyed; it can only be transformed.
Thermodynamics: heat, work, and entropy
These developments led to thermodynamics, the science of heat, work, temperature, and energy transformations. William Thomson (Lord Kelvin) helped unify the ideas, while Rudolf Clausius gave thermodynamics a rigorous formulation and helped develop the concept of entropy.
Later figures, including Walther Nernst, extended thermodynamics further. By the late 19th century, energy had become a universal organizing principle across physics, not just a concept from mechanics.
Relativity and quantum theory expand the concept
In 1905, Albert Einstein showed through mass–energy equivalence that mass and energy are deeply linked, often summarized by the relation associated with E = mc².
Around the same period, quantum theory revealed that in many microscopic systems, energy is not continuous but occurs in discrete quantized levels. That pushed the concept beyond classical mechanics and thermodynamics into modern atomic and quantum physics.

Noether’s theorem and the modern view of energy
In 1918, Emmy Noether showed that conservation laws are linked to symmetries. If the laws of physics do not change with time, then energy is conserved—conservation of energy is tied to time-translation symmetry.
Later, Richard Feynman expressed a clear modern view: energy is a numerical quantity that remains unchanged through all the transformations nature undergoes. It is not a visible substance; it is an abstract but exact conserved quantity.
Short Summary
Energy began as a philosophical idea in Aristotle, moved toward a mechanical concept in Leibniz’s vis viva, was named and clarified in 19th-century physics through Young, Coriolis, Rankine, Joule, Mayer, Helmholtz, Kelvin, and Clausius, and was later deepened by Einstein, quantum theory, and Noether. Today it is understood as one of the most basic conserved quantities in all of physics.
Simple Definition
Energy is the ability to do work or make something happen or change.
Science Definition
Energy is a scalar, conserved physical quantity characterizing capacity for change, work, or heat transfer; transformable but constant in total within isolated systems.
Reference Links
- Aristotle / energeia / actuality: Britannica (Aristotle), Britannica (Actuality)
- Thomas Young and early modern use of “energy”: Archive.org (Young lectures), Young (1807) PDF, Wikipedia (Thomas Young)
- History of energy overview: Wikipedia (History of energy)
- Daniel Bernoulli and Hydrodynamica: Britannica (Daniel Bernoulli), Britannica (Hydrodynamica)
- Émilie du Châtelet and the vis viva debate: Wikipedia (Émilie du Châtelet)
- Coriolis, work, and kinetic energy: Britannica (Coriolis), Britannica (Du calcul de l’effet des machines)
- Rankine and potential energy: MacTutor (Rankine), OSTI (Rankine ref)
- Work (force through distance): NASA (Work)
- Lavoisier and conservation of mass: Britannica (Lavoisier), Britannica (Conservation of mass)
- Joule and mechanical equivalent of heat: Britannica (Joule), Britannica (Joule’s law)
- Mayer and Helmholtz: Britannica (Mayer), Britannica (Helmholtz)
- Thermodynamics / Kelvin / Clausius: Britannica (Thermodynamics), Britannica (Laws of thermodynamics), Britannica (Second law), Britannica (Kelvin)
- Einstein and mass–energy equivalence: Britannica (E = mc²)
- Quantum / quantized energy levels: Britannica (Quantized level), Britannica (Bohr’s model), Britannica (Energy state)
- Noether / symmetry / conservation laws: Britannica (Noether video), Wikipedia (Noether’s theorem)
- Feynman on conservation of energy: Feynman Lectures (Energy)
